 a group the elements become more reactive this is because the atomic radius increases the. The covalent radii of these covalent molecules are often regarded as atomic radii. (b) Elements of group 17, in decreasing order of reactivity. Note: It is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Thus, we can conclude that the answer to this question is option (A). > Atomic radii decrease with increase in atomic number because within the same period the number of shells of electrons do not increase but positive charge on nucleus gradually increases. In a particular period of the periodic table. 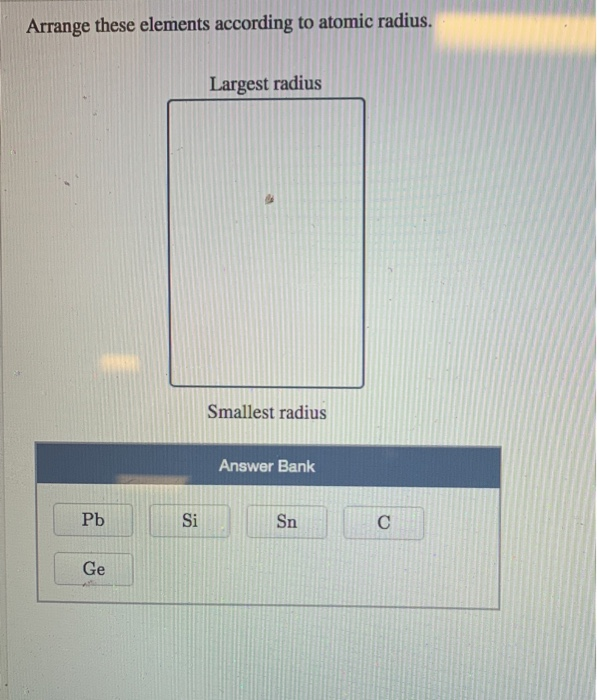
So, fluorine will have the smallest atomic radii among them three and oxygen will have smaller atomic radii than carbon’s. Hint: The atomic radius shows the distance between the centre of the atom and the valence orbital of the atom. K and Ca are of same period but K is in group 1 and Ca is in group 2. C, O and F have atomic numbers of 6, 8 and 9 respectively. Atomic size gradually decreases from left to right across a period of elements. > C, O and F are elements of the same period and in the same period atomic radius decreases with increase in atomic number. So, its atomic radius will be more than second period elements and less than bromine. 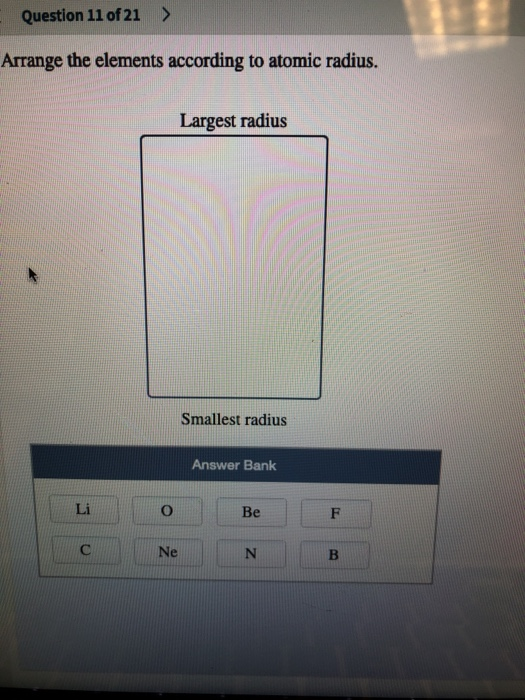
> Now, chlorine is a third period element and hence it will have a shell of electrons more than the second period elements. So, bromine will have an extra shell of electrons and hence its atomic radius will be biggest amongst all. > Bromine is an element of the fourth period while all the other elements given are of second and third period. 
Let’s see how their atomic radii will be related with each other. The atomic radius of a chemical element is defined as ‘a measure of the size of its atoms, usually the mean or typical distance from the centre of the nucleus to the boundary of the valence shell of electrons.’ The general trend is that atomic sizes increase as one moves downwards in the Periodic Table of the Elements, as electrons fill outer electron shells. Let us first try to understand what the atomic radius of an element really is before moving on to try and understand the trends of atomic sizes in the Periodic Table. In a particular period of the periodic table, atomic radius of elements decreases with increase in atomic number. Hint: The atomic radius shows the distance between the centre of the atom and the valence orbital of the atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
